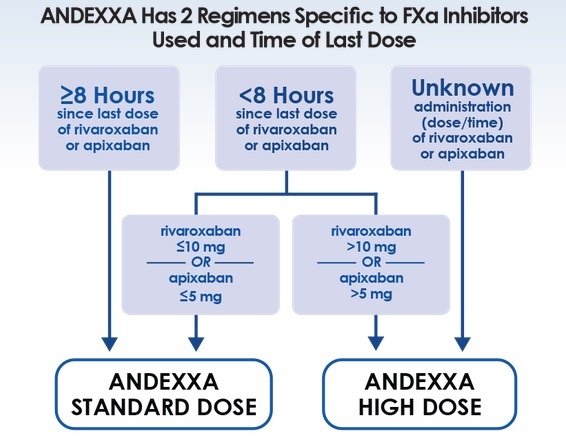
Andexanet alfa is FDA approved for the reversal of factor Xa (FXa) inhibitor-associated bleeding. 4F-PCCs were developed and approved specifically to replace depleted coagulation factors in patients receiving warfarin. However, 4F-PCCs are not the standard of care for direct oral anticoagulant (DOAC)-associated bleeding, nor are they Food and Drug Administration (FDA)-approved for this indication. state that no studies compare andexanet alfa to the standard of care, four-factor prothrombin complex concentrates (4F-PCCs). However, many statements in the publication require further clarification, which I address below. 
I read with interest the article evaluating ANDEXXA (coagulation factor Xa, inactivated-zhzo andexanet alfa ) for formulary addition by Peled et al.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
